Expo
view channel
view channel
view channel
view channel
view channel
view channel
view channel
RadiographyMRIUltrasound
General/Advanced ImagingImaging ITIndustry News
Events

- New Contrast Agent Enables Low-Dose X-Ray Joint Imaging
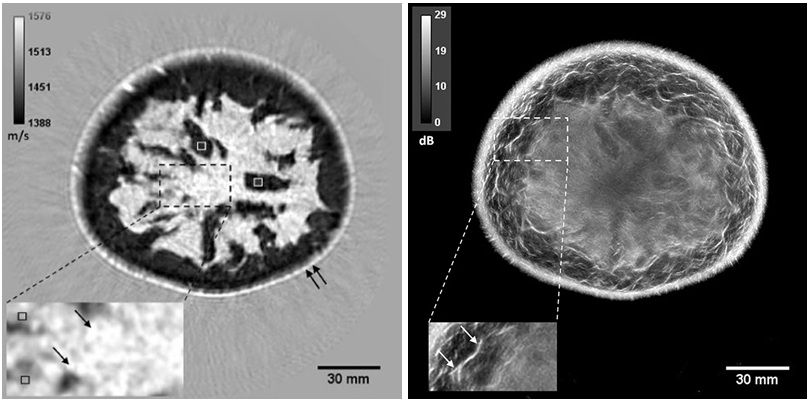
- AI Boosts Breast Cancer Detection and Cuts Screening Workload
- AI Tool Predicts Breast Cancer Risk Years Ahead Using Routine Mammograms
- Routine Mammograms Could Predict Future Cardiovascular Disease in Women
- AI Detects Early Signs of Aging from Chest X-Rays
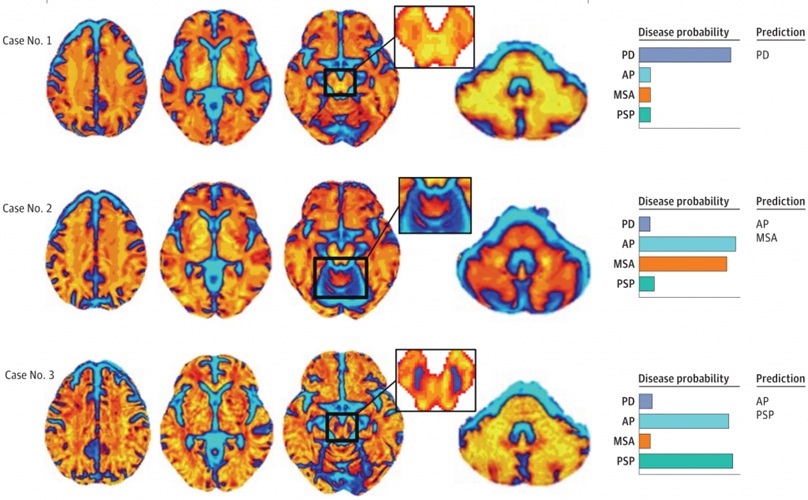
- MRI-Based AI Tool Supports Differentiation of Parkinsonian Syndromes
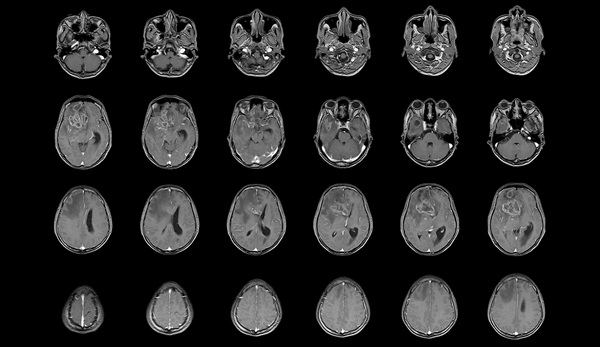
- MRI-Derived Biomarker Improves Risk Stratification in Glioblastoma
- Combined Imaging Approach Identifies Cause of Heart Attack without Coronary Blockage
- Advanced MRI System Detects Impaired Cardiac Oxygen Use in Minutes
- AI-Enhanced MRI Improves Image Quality in Arrhythmia Patients
- PET Tracer Enables Noninvasive Measurement of Beta Cell Mass
- New Imaging Tool Sheds Light on Tumor Fat Metabolism
- Radiopharmaceutical Molecule Marker to Improve Choice of Bladder Cancer Therapies
- Cancer “Flashlight” Shows Who Can Benefit from Targeted Treatments
- PET Imaging of Inflammation Predicts Recovery and Guides Therapy After Heart Attack
- New Ultrasound AI Tool Supports Rapid Prenatal Assessment
- New Consensus Standardizes Ultrasound-Based Fatty Liver Assessment
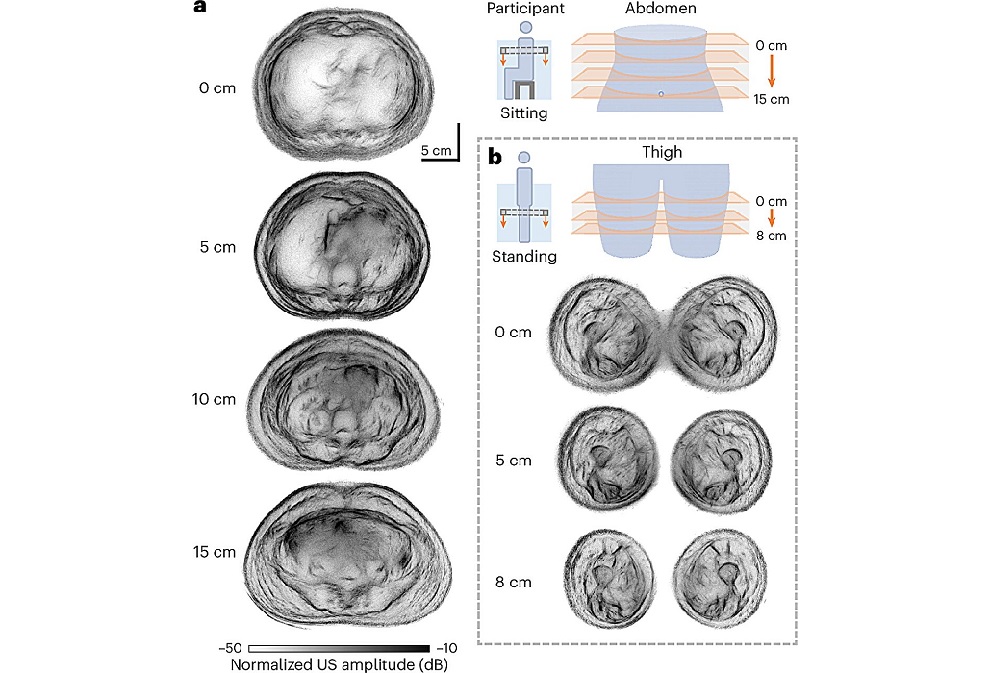
- Groundbreaking Technology to Enhance Precision in Emergency and Critical Care
- Reusable Gel Pad Made from Tamarind Seed Could Transform Ultrasound Examinations
- AI Model Accurately Detects Placenta Accreta in Pregnancy Before Delivery
- New Breast Imaging Viewer Unifies Modalities and Enhances Clinical Workflow
- Hybrid AI System Improves Early Lung Cancer Detection on CT
- Radiomics Analysis of CT Scans Enhances Evaluation of Sarcoidosis
- AI Tool Predicts Side Effects from Lung Cancer Treatment
- AI Tool Offers Prognosis for Patients with Head and Neck Cancer
- New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
- Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
- AI-Based Mammography Triage Software Helps Dramatically Improve Interpretation Process
- Artificial Intelligence (AI) Program Accurately Predicts Lung Cancer Risk from CT Images
- Image Management Platform Streamlines Treatment Plans
- GE HealthCare and NVIDIA Collaboration to Reimagine Diagnostic Imaging
- Patient-Specific 3D-Printed Phantoms Transform CT Imaging
- Siemens and Sectra Collaborate on Enhancing Radiology Workflows
- Bracco Diagnostics and ColoWatch Partner to Expand Availability CRC Screening Tests Using Virtual Colonoscopy
- Mindray Partners with TeleRay to Streamline Ultrasound Delivery

 Expo
Expo
- New Contrast Agent Enables Low-Dose X-Ray Joint Imaging
- AI Boosts Breast Cancer Detection and Cuts Screening Workload
- AI Tool Predicts Breast Cancer Risk Years Ahead Using Routine Mammograms
- Routine Mammograms Could Predict Future Cardiovascular Disease in Women
- AI Detects Early Signs of Aging from Chest X-Rays
- MRI-Based AI Tool Supports Differentiation of Parkinsonian Syndromes
- MRI-Derived Biomarker Improves Risk Stratification in Glioblastoma
- Combined Imaging Approach Identifies Cause of Heart Attack without Coronary Blockage
- Advanced MRI System Detects Impaired Cardiac Oxygen Use in Minutes
- AI-Enhanced MRI Improves Image Quality in Arrhythmia Patients
- PET Tracer Enables Noninvasive Measurement of Beta Cell Mass
- New Imaging Tool Sheds Light on Tumor Fat Metabolism
- Radiopharmaceutical Molecule Marker to Improve Choice of Bladder Cancer Therapies
- Cancer “Flashlight” Shows Who Can Benefit from Targeted Treatments
- PET Imaging of Inflammation Predicts Recovery and Guides Therapy After Heart Attack
- New Ultrasound AI Tool Supports Rapid Prenatal Assessment
- New Consensus Standardizes Ultrasound-Based Fatty Liver Assessment
- Groundbreaking Technology to Enhance Precision in Emergency and Critical Care
- Reusable Gel Pad Made from Tamarind Seed Could Transform Ultrasound Examinations
- AI Model Accurately Detects Placenta Accreta in Pregnancy Before Delivery
- New Breast Imaging Viewer Unifies Modalities and Enhances Clinical Workflow
- Hybrid AI System Improves Early Lung Cancer Detection on CT
- Radiomics Analysis of CT Scans Enhances Evaluation of Sarcoidosis
- AI Tool Predicts Side Effects from Lung Cancer Treatment
- AI Tool Offers Prognosis for Patients with Head and Neck Cancer
- New Google Cloud Medical Imaging Suite Makes Imaging Healthcare Data More Accessible
- Global AI in Medical Diagnostics Market to Be Driven by Demand for Image Recognition in Radiology
- AI-Based Mammography Triage Software Helps Dramatically Improve Interpretation Process
- Artificial Intelligence (AI) Program Accurately Predicts Lung Cancer Risk from CT Images
- Image Management Platform Streamlines Treatment Plans
- GE HealthCare and NVIDIA Collaboration to Reimagine Diagnostic Imaging
- Patient-Specific 3D-Printed Phantoms Transform CT Imaging
- Siemens and Sectra Collaborate on Enhancing Radiology Workflows
- Bracco Diagnostics and ColoWatch Partner to Expand Availability CRC Screening Tests Using Virtual Colonoscopy
- Mindray Partners with TeleRay to Streamline Ultrasound Delivery